 
So, the product and reactants of chemical equations should have an equal number of atoms on both sides. Here is How to Use Balancing Equations Calculator Chemistry. Besides, in this article, we will learn how to balance chemical equations?Īs per the law of conservation of mass, “atom can neither be created nor can be destroyed”. Instructions to Use Balancing Equations Calculator Chemistry. In addition, we connect these two sides with an arrow leading from the left to the right, symbolizing the reaction. Moreover, in a chemical reaction, the reactants are written on the left side and products are written on the right side. Basically, the symbols are the letter that represents an element. Step 4: Check that all the atoms balance and make sure that all coefficients are in the lowest-possible ratio.Symbolic representation of a chemical reaction is referred to as a chemical equation. Change the coefficient in front of the O 2 to 3 to give eight O on the left. Step 3: The C and H are still balanced, and now there are six O on the left and eight on the right. We multiply both sides of the equation through by two. Therefore, we will just balance the reduction part of the reaction. In the example question, the oxidation part of the reaction in terms of Fe atoms is already balanced. Step 2: When we try to balance the oxygen, we find three on the left and four on the right. In the third step of balancing redox reactions by half-reaction method, we will balance the atoms present in each half of the reaction except O and H atoms. Use uppercase for the first character in the element and lowercase for the second. The remaining values will automatically be calculated. The reactants and products, along with their coefficients will appear above. There are four H on the left and two on the right, so we place the coefficient of 2 in front of the H 2O on the right To perform a stoichiometric calculation, enter an equation of a chemical reaction and press the Start button. Step 1: Using the CHO technique, we start with carbon, one on each side, so carbon is balanced. This will give an even number on both sides and make the equation easier to balance. If you have an even number of a certain element on one side of the equation and an odd number of the same element on the other side of the equation, multiply both sides of the equation through by the coefficient of 2. Step 2: To obtain a balanced equation, now click on 'Balance'. Rule 4: Balancing chemical equations using the even/odd technique. Step 1: First, enter the chemical formula in the text box. In this lesson, we will look at some examples of applying Rule 5Balancing chemical equations containing polyatomic ions Solid potassium chlorate, KClO 3, decomposes to form solid potassium chloride and diatomic oxygen gas. 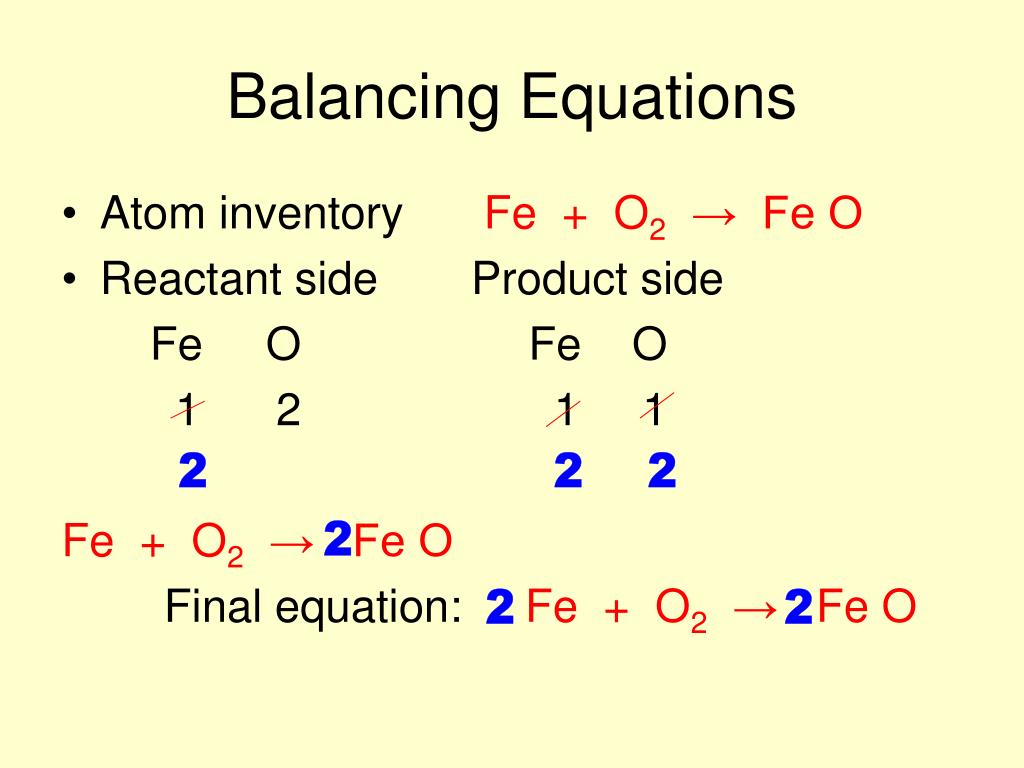
Rule 4Balancing chemical equations using the even/odd technique Write a balanced equation describing each of the following chemical reactions. Second, change the coefficient of one of the substances. Rule 3Balancing chemical equations using the CHO technique These are the steps: First, count the atoms on each side. Rule 2Balancing chemical equations using the two’s and three’s technique General Rules for balancing chemical equations Rule 1Balancing chemical equations using the one’s and two’s technique. Scroll down the page for more examples and solutions. The following figure gives some hints on how to balance chemical equations. In this lesson, we will elaborate on Rule 4, which is balancing chemical equations using the even/odd technique. There are five common rules (or techniques) that can help us to balance chemical equations. This means that chemical equations need to be balanced before they can be used for calculations. The sum of atoms before reaction = the sum of atoms after reaction Related: If you want to know the percent composition of molecules or compounds then you can use our percent comp calculator. Our calculator provides steps so that you will get more familiar with the steps. Use uppercase for the first character in the element and lowercase for the second character. If you want to know the step-by-step process of balancing a chemical equation then you can directly click on it. 
There is only one atom of carbon on the left-hand side, but six on the right-hand side. Here, both carbon and hydrogen fit this requirement. 
The balanced equation will be calculated along with the oxidation states of each element and the oxidizing and reduction agents. The first step to balancing chemical equations is to focus on elements that only appear once on each side of the equation. Therefore, in a chemical reaction the sum of atoms before a reaction is equal to the sum of atoms after a reaction. Enter an equation of a redox chemical reaction and press the Balance button. Atoms can neither be destroyed nor created during a simple chemical reaction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
